|
The valence electrons occupy higher levels due to the increasing quantum number (n). Because electrical resistivity is typically measured only for solids and liquids, the gaseous elements do not appear in part (a).\( \newcommand\): Periodic Table showing Atomic Radius Trendĭ own a group, atomic radius increases. Ans: Hint:Electronegativity is defined as the tendency of the. On the periodic table, electronegativity generally increases as you move from left to right across a period and decreases as you move down a group. The organization of the periodic table reveals many important trends in the physical properties of the elements Atomic size Electronegativity Electron. Arrange these elements according to electronegativity: F, C, Ga, K, Cs. increasing electronegativity Arrange the following elements in order of decreasing electronegativity. How would you arrange these elements in order of decreasing electronegativity: N, Al, Na, P. Arrange these elements in order of decreasing first ionization energy: Cl, S, Sn, Pb. Order the following elements: S, Si, Al, Mg, and Ca A. So night prism of season, sodium and aluminum CCM are even so we had. 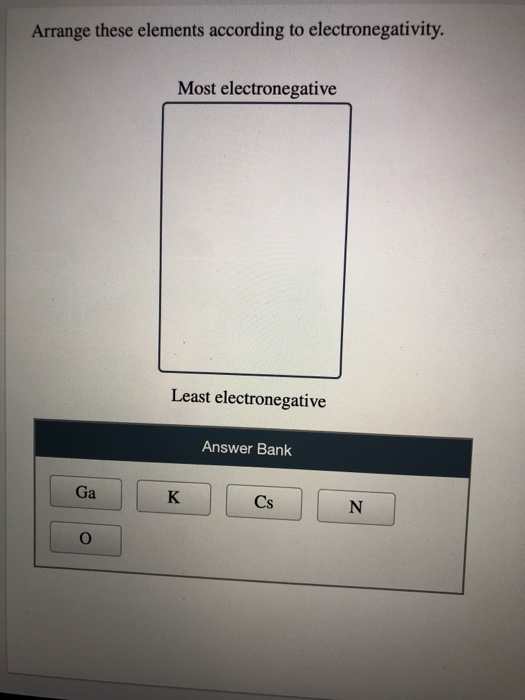
Low electronegativity values (≤ about 2.2) correlate with low resistivities (metals). Arrange these elements in order of decreasing electronegativity: Rank from largest to smallest. So you can hear in this question we have some elements. View solution > Arrange F, C, O, N in the decreasing order electronegativity. The electronegativity of the following elements increase in the order : Medium. Arrange Cs, F, and Cl in order of increasing electronegativity. Study with Quizlet and memorize flashcards containing terms like What information is needed to determine the energy of an electron in a many-electron atom Select all that apply., What information is most important in determining the size of an orbital, What information is needed to determine the orientation of an orbital and more. (b) A plot of Pauling electronegativities for a like set of elements shows that high electronegativity values (≥ about 2.2) correlate with high electrical resistivities (insulators). Click hereto get an answer to your question Arrange the following elements in the increasing order of electronegativity. If the molecules are ionic, draw the lewis dot structure and Lewis or dot a nay to the arranged in is Draw a Lewis Dot Structure for the following elements. 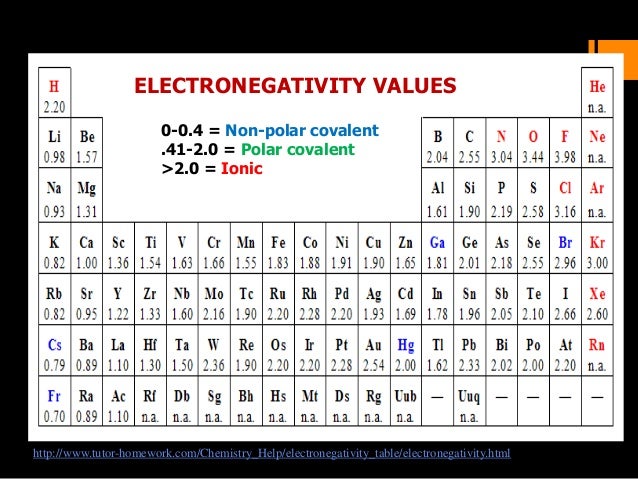
Ionization Energy C) Ionic Radius D) Electron Affinity E) Electronegativity. 
(a) A plot of electrical resistivity (measured resistivity to electron flow) at or near room temperature shows that substances with high resistivity (little to no measured electron flow) are electrical insulators, whereas substances with low resistivity (high measured electron flow) are metals. Rb, In, Sn, Sb, As Arrange the atoms according to ionization energy (five. \( \newcommand\): Three-Dimensional Plots Demonstrating the Relationship between Electronegativity and the Metallic/Nonmetallic Character of the Elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
